Clinical and Anatomic Outcomes of 3-Piece Poly IOL Rescue and Needle-Assisted Transconjunctival Intrascleral Lens Fixation
Christian Curran, MD
Murtaza K. Adam, MD
Article published in Journal of VitreoRetinal Diseases by America Society of Retinal Specialists (ASRS)
Abstract
Purpose: To report the clinical and anatomic outcomes of a single-stage rescue and sutureless 30-gauge needle-assisted transconjunctival intrascleral fixation of dislocated 3-piece rigid poly(methyl methacrylate) (PMMA) intraocular lenses (IOLs).
Methods: This retrospective noncomparative single-surgeon interventional study comprised eyes that had concurrent 23- or 25-gauge pars plana vitrectomy and IOL rescue with sutureless transconjunctival needle-assisted flanged haptic intrascleral fixation. Lamellar scleral dissection for haptic fixation was performed 3 mm posterior to the surgical limbus with 30-gauge needles. Primary endpoints included visual acuity (VA), IOL tilt measured with ultrasound biomicroscopy (UBM), and postoperative complications.
Results: The study evaluated 25 eyes of 24 patients. The IOL was successfully refixated in 24 of the 25 eyes.
The mean preoperative logMAR VA of 1.21 ± 0.79 SD (median 1.3; Snellen equivalent 20/400) improved to 0.28 ± 0.35 (median 0.14; Snellen equivalent 20/30) postoperatively (P < .0001). The mean IOL tilt measured by UBM (n = 7) was 3.79 ± 3.60 degrees. The mean postoperative follow-up was 348 ± 284 days (range, 7-979 days). Postoperative complications included self-clearing vitreous hemorrhage (n = 9), retinal detachment (RD) (n = 1), cystoid macular edema (n = 3), and corneal edema (n = 3). Three eyes (13%) required additional surgery, 1 for RD and 2 for delayed haptic slippage and secondary IOL tilt causing irregular astigmatism.
Conclusions: Intrascleral needle-assisted fixation of dislocated or mobile 3-piece PMMA IOLs is an effective, safe method to restore VA.
Introduction
Dislocated or mobile 3-piece rigid poly(methyl methacrylate) (PMMA) intraocular lenses (IOLs) present a challenge for the ophthalmic surgeon. Unlike foldable hydrogel, silicone, or acrylic IOLs, PMMA IOL optics are not flexible, cannot be cut in half, and typically cannot be inserted or removed through a corneal or scleral tunnel incision smaller than 6.0 mm in width. Although foldable IOLs have been available since the 1980s, rigid 3-piece PMMA IOLs were still implanted by cataract surgeons well into the 1990s. After decades of stability, capsule incompetence from previous surgery, Soemmerring ring growth, and zonulopathies of various etiologies can place PMMA IOLs
at risk for mobility or dislocation.
Numerous surgical options are available for patients with dislocated 3-piece IOLs. Posterior chamber rescue of IOLs with pars plana vitrectomy (PPV) is a well-established, safe, and effective methodology when combined with IOL exchange1,2 or refixation of the primary IOL to the capsule, iris, or sclera.3 Capsule refixation is a less than ideal option because an underlying zonulopathy or capsule defect is the source of IOL instability. Iris fixation can be complicated by secondary pigment dispersion, intraocular hemorrhage, and elevated intraocular pressure (IOP).4–6 IOL exchange with an anterior chamber IOL (ACIOL) is a well-established technique; however, precise IOL sizing, corneal endothelial cell loss (ECL), and the need for a large incision to perform the exchange convey clear surgical
disadvantages.
When addressing dislocated 3-piece PMMA IOLs, IOL rescue with refixation to the sclera has several advantages. With placement of the IOL in the retropupillary plane, the theoretical risk for corneal ECL and iris chafing is lower. Furthermore, by avoiding IOL explantation via a 6.0 mm corneal or sclerocorneal wound, intraoperative hypotony and postoperative astigmatism can be minimized.
There is no consensus regarding the superiority of any given secondary fixation technique because each has unique risks and benefits.7 Previous studies reported success in rescuing 3-piece IOLs with intrascleral haptic fixation (ISHF) using scleral flaps, fibrin glue, sutures, and small-gauge trocar cannulas. 8 First described by Yamane et al,9 a sutureless needleassisted technique to fixate 3-piece IOLs is a simple, elegant approach to ISHF with minimal instrumentation. This technique has gained popularity for its efficacy in providing good centration and minimal tilt of the implanted IOL, excellent best-corrected visual acuity (BCVA), and low complication rates. However, a systematic Medline/PubMed literature search found this approach has only been described using modern foldable IOLs in the context of IOL exchanges or primary implantation in eyes lacking capsule support.
Here, we describe a new technique and the retrospective outcomes of a sutureless needle-assisted method to rescue 3-piece PMMA IOLs via ISHF.
Methods
In this retrospective noncomparative interventional study, the charts of all patients who had combined PPV and IOL repositioning with scleral fixation by the same surgeon (M.K.A.) between April 2018 and July 2021 were retrospectively reviewed. One patient was excluded from analysis for loss to follow-up after postoperative day 1. One patient with limited follow-up (1 week) was included because the patient had postoperative ultrasound biomicroscopy (UBM) to measure IOL tilt. Not every patient in this series had access to UBM during
the postoperative course. The following data were collected for all patients: age, sex, preoperative and postoperative Snellen VA, preoperative and postoperative IOP, surgical indications, and intraoperative and postoperative outcomes.
Patients were observed for the following postoperative complications: retinal detachment (RD), hypotony, ocular hypertension, glaucoma, iris capture, corneal edema, wound leak, choroidal hemorrhage, vitreous hemorrhage (VH), epiretinal membrane (ERM), vitreous prolapse, and the need for a reoperation. If a subclinical or clinical condition was present before the intervention, that condition was not regarded as a surgical complication unless it substantially worsened within 6 months of the procedure. All surgical complications that were included had persisted beyond the initial 1-month postoperative recovery window, arose within 6 months of surgery, or required surgical intervention.
All patients had a standardized ophthalmic examination to assess their candidacy for surgery. All patients provided informed consent before surgery. The presence of a PMMA IOL was confirmed by the intraoperative examination and apparent rigidity of the IOL optic during IOL manipulation. All included patients originally had the IOL placed in the capsular bag or ciliary sulcus.
Surgical Technique
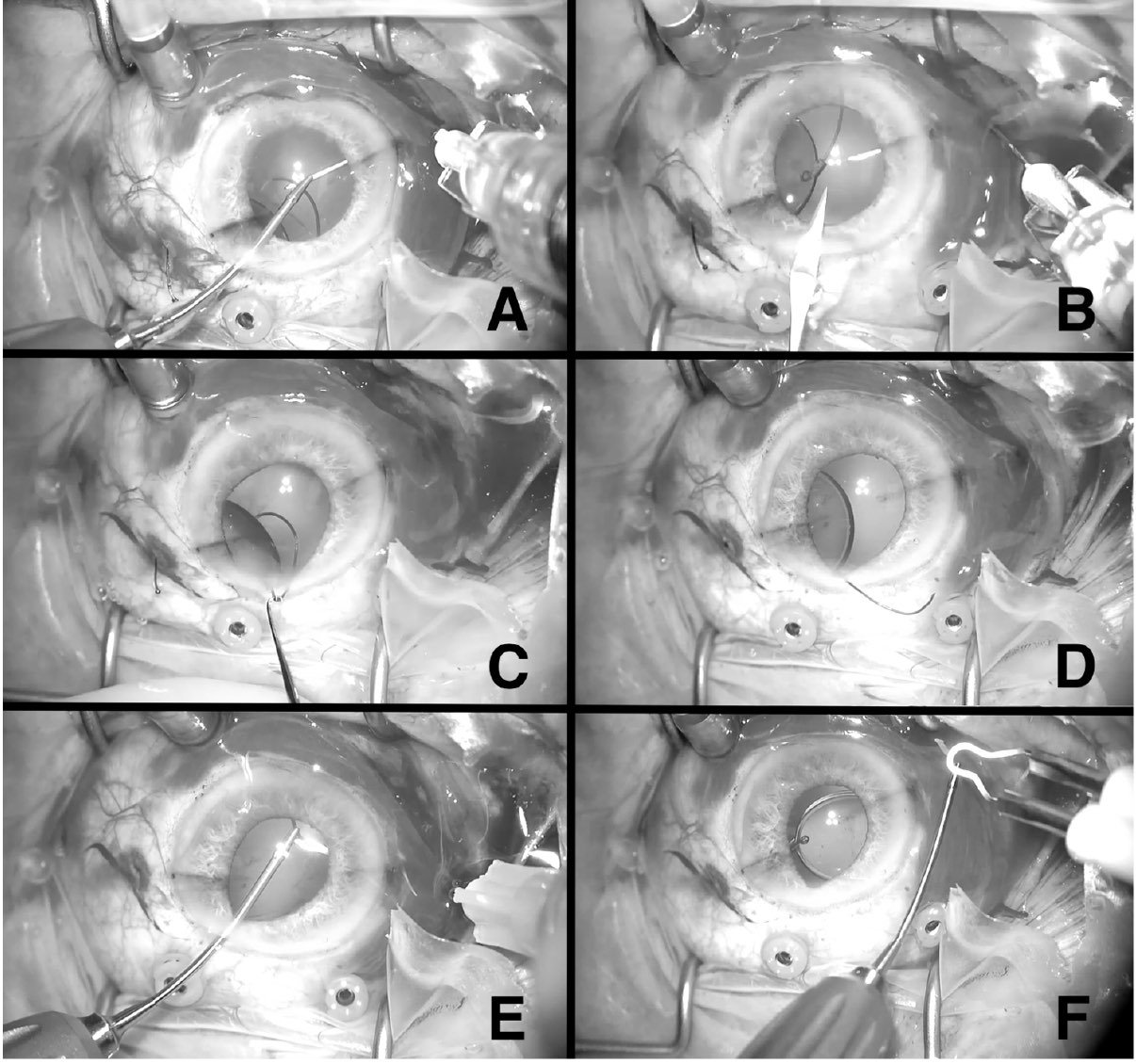
In each case, a standard 3-port 23-gauge or 25-gauge PPV was concurrently performed with IOL rescue and needle-assisted ISHF. Figure 1 shows key portions of the procedure. After a complete vitrectomy was performed and an intracameral viscoelastic was injected, the PMMA IOL was elevated into the anterior
chamber with an intraocular forceps. Paracenteses were made at the superotemporal and superonasal limbus, and a
3 mm lamellar clear corneal incision (CCI) was created at the superior limbus with a keratome blade. If associated capsule, Soemmerring ring, or both were adherent to the IOL, they were removed through the superior corneal wound with the assistance of an intraocular forceps and scissors.
After a toric marker was used to delineate the horizontal meridian of the cornea centered on the central visual axis, scleral markings were made 3 mm posterior to the limbus 180 degrees apart. The trailing haptic of the IOL was then externalized from the eye via the superior corneal wound. Next, 2 thinwalled 30-gauge needles (TSK Lab) were introduced into the vitreous cavity via opposing beveled sclerotomies initiated at the scleral markings. Using an intraocular forceps and hypodermic needles as originally described by Yamane et al,9 the leading and trailing haptics were docked into the 2 needles, first externalizing the leading haptic and then the trailing haptic. The
creation of the superior lamellar corneal incision allows for a more ergonomic angle to dock the trailing haptic into the second needle. Using thermal cautery, terminal bulbs were formed at the end of each externalized haptic and the haptics were placed into the subconjunctival space to secure them within their respective sclerotomies.
Imaging
In eyes in which UBM was performed, the tilt of the scleralfixated IOL was measured using the EyeCubed 40 MHz machine (Ellex). The images depicted the vertical (0-degree), oblique (45-degree), and horizontal (90-degree) angles of the final IOL placement. Optic tilt was measured using the most common method, with minor modifications.10,11 The established methodology for analyzing IOL tilt via UBM imagery includes the following steps:
- A straight line is drawn between the center of the 2 sides of the iris at the hyperlucent pigmented epithelium on the posterior aspect of the iris plane. This defines the angle of reference.
- A line is generated tangent to the curve of the IOL, which is the angle of incidence. The angle between them is then measured to establish tilt.
- Absolute measurements are taken by drawing lines perpendicular to the angle of reference from the medial edge of both pupillary margins and extending these lines to the line of incidence. If the difference between the 2 sides was 0.1 mm or more, the IOL was considered to be tilted.10–13
The vertical, oblique, and horizontal angles of imagery were combined into a mean composite measurement, which was then recorded. All images were analyzed using Adobe Illustrator (version 25.2.3, Adobe Inc), which allowed for precise elliptical modeling of the IOL contour to mathematically draw tangent lines and measure angles (Figure 2). Measurements of absolute distance were made by analyzing these image files with ImageJ software (version 1.53a, National Institutes of Health). Mean values are ± SD.
Statistical Analysis
Snellen VA was converted to logMAR notation using a previously published technique14 and Excel software (version 16.49, Microsoft Corp). The nonparametric Mann-Whitney U test was used to compare preoperative and postoperative visual outcomes, with P < .05 considered the threshold of significance. Statistical analyses were performed using Prism 8 software (version 8.4.3, GraphPad Software, LLC).
Results
This study included 25 eyes of 24 patients with a mean age on the date of procedure of 76 ± 9.5 years. Table 1 shows the patients’ demographics. In all eyes but 1, the indication for surgery was a dislocated IOL; that 1 patient was monocular and had surgery for visually significant pseudophacodonesis. The mean logMAR BCVA before the intervention was 1.21 ± 0.79, with a range of 0.0 to 2.7 (Snellen equivalent 20/320; range, 20/20-light perception). Previous procedures and diagnoses included RD, scleral buckle, PPV, glaucoma, and concurrent cystoid macular edema (CME). The mean follow-up was 348 ± 284 days.
Intervention Results
The PMMA IOL was successfully repositioned in 24 (96%) of 25 eyes. In addition to ISHF of the PMMA IOL, all eyes had a 23-gauge or 25-gauge PPV, 10 (40%) had therapeutic debulking

of a Soemmerring ring, 3 (12%) had ERM peeling, and 3 (12%) had endolaser treatment for peripheral RD, retinal tear, or detachment prophylaxis. The logMAR VA significantly improved to 0.28 ± 0.35 (median 0.14; Snellen equivalent 20/30) (Mann Whitney U = 78; n1 = 25; n2 = 25; P < .0001).
UBM measurements of IOL tilt were obtained for 6 patients (7 eyes). The mean tilt angle was 3.51 ± 3.77 degrees (range, 0-15.18 degrees) with a mean absolute tilt of 0.20 ± 0.28 mm (range, 0-0.93 mm). The mean best measured logMAR VA for patients having UBM was 0.25 ± 0.38. Four eyes (57%) had a mean absolute IOL tilt of less than 100 μm. There was no significant difference in the best postoperative VA between patients having UBM and those not having UBM (median 0.08 vs 0.24) (Mann-Whitney U = 44; n1 = 7; n2 = 18; P = .2614). No significant correlation was found between the angle of tilt observed on UBM and postoperative VA (Spearman r test: rs = 0.26, R2 = 0.067; P = .5905, 2-tailed). No significant difference was found between the final VA of patients with preexisting glaucoma and those without it (median logMAR 0.22 [Snellen equivalent 20/30] vs 0.18 [Snellen equivalent 20/30]) (Mann-Whitney U = 53.5; n1 = 7; n2 = 18; P = .8423).
Complications
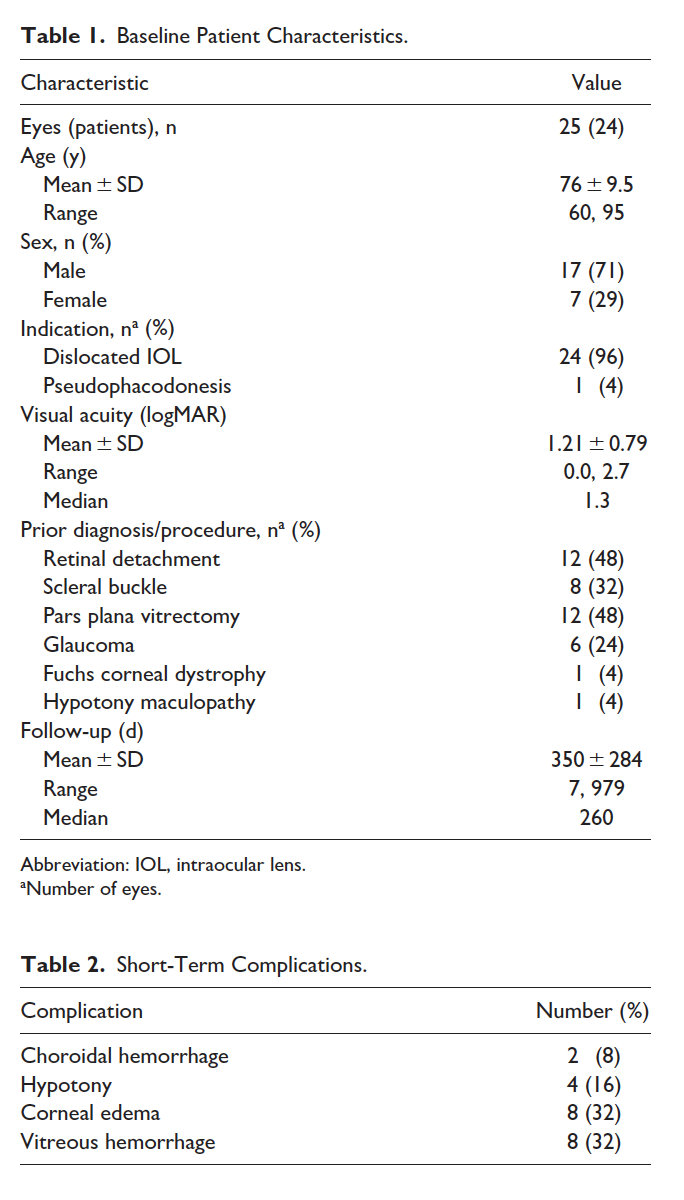
Table 2 lists the observed postoperative complications, which are generally regarded as part of the acute recovery phase; that is, short-term, self-resolving complications. The most common complications were corneal edema and VH followed by hypotony and choroidal hemorrhage.

During the study period, 22 (88%) of 25 eyes (21 of 24 patients) did not require a reoperation. Two patients developed irregular astigmatism as a result of delayed decentration caused by slippage of 1 of the haptics within the associated scleral fixation site; this was first noted at the 3-month follow-up appointment. In both astigmatism cases, the BCVA was 20/60 after PMMA IOL refixation using the ISHF technique. It was assumed that the irregular astigmatism from IOL tilt did not allow spectacle correction to achieve visual potential for these eyes. Because of the office location for these patients, UBM data were not obtained in these eyes. Both patients elected for an exchange with an aspheric acrylic 3-piece IOL (CT Lucia 602, Zeiss Inc) and recovered fully, achieving 20/20 VA.
One patient with a history of RD repaired with vitrectomy, 360-degree laser, and gas approximately 25 years previously developed macula-involving RD 3 weeks after IOL rescue with ISHF. RD repair with repeat vitrectomy and membrane peeling, laser, peripheral iridotomy, and gas tamponade was performed. After 11 months of postoperative follow-up, the patient had a VA of 20/30 with ongoing topical treatment for ocular hypertension and CME, which were not present after his ISHF surgery alone.
Finally, 1 patient had a complex history of traumatic glaucoma status after trabeculectomy complicated by bleb-associated endophthalmitis and subsequent chronic hypotony. This complex eye had vitrectomy for the endophthalmitis and bleb revision to address hypotony before developing a dislocated IOL. After IOL rescue with ISHF, the patient had significant IOL tilt and severe corneal edema. The preoperative VA of 20/400 marginally worsened to counting fingers, and the patient elected to be observed rather than to pursue further surgery.
Conclusions
To our knowledge, this is the largest series of PMMA IOL rescues to date. Our results support previous findings that sutureless ISHF avoids several disadvantages of the sutured and glued techniques.
Sutured haptic fixation can be complicated by dysfunction of the suture itself, leading to inflammation, infection, or haptic slippage, which can further lead to IOL decentration and dislocation. 15–19 Attempts to mitigate these complications with polytetrafluoroethylene (Gore-Tex) sutures have shown acceptable refractive outcomes20 but have also introduced new complications, such as granulation tissue formation into the ciliary body and sclera, IOL opacification, eyelet fractures with subsequent IOL dislocation, persistent corneal edema, and erosion of the suture through the conjunctiva.21–25 Sutureless ISHF using fibrin glue has relatively lower rates of postoperative complications; however, glue-assisted techniques generally use the formation of a scleral flap,8 which represents substantial surgical dissection. Although both glued and sutured techniques remain important methods of intrascleral fixation, our findings suggest that the sutureless flanged ISHF technique offers equivalent outcomes while avoiding the risk for complications and increased surgical manipulation inherent to these other techniques.
In addition, the double-needle technique used in our study may offer advantages over another common method of ISHF, the trocar-assisted technique. Trocar-assisted ISHF involves relatively larger sclerotomies, more instrumentation, and greater cost. The larger tunnels also require robust flanging because they present a significantly greater risk for slippage in unflanged cases than in flanged cases (18.5% vs 6.6%; P < .0001).26,27 The use of a 30-gauge sclerotomy to fixate a flanged haptic theoretically confers better long-term IOL stability.
The major difference between previously discussed doubleneedle techniques and the method used in our study is the creation of a 3 mm lamellar CCI made superiorly with externalization of the trailing haptic before needle docking of the trailing haptic. This incision provides a more ergonomic angle when docking the trailing haptic into the second 30-gauge needle, improving intraoperative maneuvering of the trailing haptic and comfort to the surgeon. This is especially valuable in the case of PMMA IOLs, where the haptics can be prone to crimping or breakage. Although 27-gauge trocar-assisted flanged haptic scleral fixation may be advantageous to vitreoretinal surgeons more familiar with this surgical method, the double-needle technique is cost-effective and simple. The modification that we propose augments those qualities without increasing the risk for large incision–related complications such as wound leak and endophthalmitis.
The results of our study are in keeping with or slightly better than those in previous reports. UBM imaging from 7 eyes (6 patients) showed a mean IOL tilt angle of 3.51 ± 3.77 degrees (range, 0-15.18 degrees) compared with the mean reported by Yamane et al9 (3.4 ± 2.5 degrees), that reported for sutured fixation (4.43 ± 3.02 degrees or greater),15,25 and that reported for glued scleral fixation (3.2 ± 2.7 degrees and 2.9 ± 2.6 degrees in the horizontal axis and vertical axis, respectively).28 In our study, the mean absolute tilt measured 0.20 ± 0.28 mm (range, 0-0.93 mm) on UBM and the mean logMAR BCVA in the patients who had UBM was 0.25 ± 0.38. This is an improvement over the results of Marianelli et al,12 who reported a mean vertical tilt of 0.24 ± 0.21 mm, a mean horizontal tilt of
0.25 ± 0.19 mm, and a mean logMAR BCVA of 0.55 ± 0.32 for patients who had sutureless ISHF. The correlation between refractive tilt and lens tilt is a function of the IOL type. Spherical IOLs, such as PMMA models, have a relatively higher tolerance for tilt up to 4 degrees but a low tolerance for decentration up to 0.5 mm before inducing a significant VA change.29 In general, an absolute IOL tilt greater than 200 μm corresponds to 2 to 3 degrees of tilt. IOL tilt of such minor magnitude is common in clinical practice, has little to no effect on VA, and remains unnoticed by most patients.30,31 These data are reassuring given that PMMA IOL refixation by our modified ISHF technique provided outcomes similar to those of previously reported techniques.
The postoperative complications in this study elicit further discussion. Our rates of postoperative hypotony, VH, wound leak, iris capture, corneal edema, CME, IOL decentration, and RD are at or below those in other studies of secondary IOL fixation techniques.3,9,32–34 Rates for ocular hypertension were slightly higher than in some previous studies. Ocular hypertension was managed medically in all cases. The 1 patient who experienced RD had significant predisposing factors including previous RD, ERM, and myopic degeneration in the same eye. He was able to have successful repair with gas tamponade without the risk for opacification, as would have been the case with a hydrophilic secondary IOL, such as the Akreos AO60 (Bausch + Lomb).35 He did ultimately develop ocular hypertension and CME from the RD surgery, which was not seen after IOL refixation alone. Although this rate (4%) is higher than in the Yamane et al9 study, in which there were no case of RD in a larger cohort, our older patient
population and the presence of a previous RD in this patient increased the likelihood of this complication occurring.
In addition, other studies of ISHF reported similar rates of RD.34 Three patients required a reoperation. One patient presented with a history of hypotony maculopathy resulting from previous trauma and might have reasonably been excluded from this study because of his significant comorbidities. However, we believed that his case represented a unique surgical challenge worth addressing. On the one hand, a large 6 mm incision to remove the PMMA IOL would pose an increased risk for postoperative wound leak, endophthalmitis, and a recurrence of hypotensive maculopathy. On the other hand, ACIOL implantation and all other scleral fixation techniques would likely have led to increased iatrogenic endothelial trauma. In an eye with this number of ocular comorbidities, the risk for postoperative complications is high regardless of the IOL fixation technique selected.
The 2 patients with delayed IOL decentration caused by presumed haptic slippage had improved VA after exchange with an aspheric IOL. The improvement is notable because PMMA IOLs are spherical and therefore worsen positive corneal spherical aberrations.36 These patients both achieved 20/20 VA with an aspheric acrylic 3-piece IOL (CT Lucia 602). One reason for PMMA IOL haptic slippage may relate to the flange configuration, where extruded PMMA haptics conform to a triangular or conical flange shape when exposed to thermal cautery.37
Conversely, the polyvinylidene fluoride haptics on the CT Lucia 602 IOL flange to a more abrupt mushroom bulb shape and thus might lead to longer term intrascleral stability. Based on our results, the creation of a large terminal haptic “bulb” for IOLs with PMMA haptics may provide better mechanical resistance to intrascleral haptic slippage.
Our instances of postoperative complications ought to be understood in the context of our patients’ high rates of preexisting pathology, such as RD (48%) in the ipsilateral eye and glaucoma (24%), as well as the mean age of our patient cohort (76 ± 9.5 years). As expected, eyes that had a more complex history of pathology tended to develop more complications after this procedure.
In our study, the lack of a significant difference between the visual outcomes of patients with preexisting glaucoma and those without in the setting of IOL dislocation is also a notable finding. The benefit of this surgical intervention in glaucoma patients has to be weighed against the potential risk for worsening the underlying condition without improving VA if the loss of VA is caused by a neurosensory change rather than a refractive error. For our patients with existing glaucoma, the calculus seems to favor intervention given that their best achieved VA was not significantly different than that of patients who presented without glaucoma.
This study has limitations worth considering in the interpretation of its results. First, it evaluated a relatively small number of eyes (n = 25), which could have skewed data toward a type I or type II error. Second, all surgeries were performed by the same surgeon; thus, the results may not be generalizable. Third, UBM imaging was not performed for all patients because it was available at only 1 one of 5 clinic locations; thus, the results in the subset of patients who had UBM might may not fully represent the overall outcomes. BCVA could not be consistently assessed because of the nature of the surgical context in which these patients received care; therefore, our results represent the corrected and uncorrected best achieved VA at postoperative follow-up appointments. Further research should be directed toward elucidating indications for IOL refixation vs IOL exchange to provide the best outcomes for patients based on their presenting conditions and history. In conclusion, sutureless flanged ISHF for a dislocated 3-piece PMMA IOL by our modified Yamane technique is a simple, safe, and effective method to restore VA without the
need for IOL explantation through a large incision. Our technique for rescuing a dislocated 3-piece PMMA IOL with flanged ISHF led to a significant improvement in VA, low longterm complication rates and, on average, excellent angulation when viewed under UBM.
Authors’ Note
Presented at the annual meetings of the American Academy of Ophthalmology (virtual meeting), November 2020, and the American Society of Retina Specialists Annual Meeting, Chicago, IL, USA, August 2019.
Ethical Approval
This retrospective study was conducted with approval by the Rocky Vista University Institutional Review Board (#2019-0106) and was in accordance with the Declaration of Helsinki and all applicable US Health Insurance Portability and Accountability Act regulations.
Statement of Informed Consent
Informed consent was obtained prior to performing the procedure, including permission for publication of all photographs and images included herein.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Adam is a consultant to Genentech, Apellis, Iveric Bio, RegenxBio, and Allergan and a speaker for Genentech and Regeneron. Dr. Curran has no competing interests to report.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD
Christian Curran orcid.org/0000-0002-2534-9482v
Citation
Original posting: article
- Curran C, Adam MK. Clinical and Anatomic Outcomes of 3-Piece Poly(methyl methacrylate) Intraocular Lens Rescue and Needle-Assisted Transconjunctival Intrascleral Lens Fixation. Journal of VitreoRetinal Diseases. 2023;7(5):404-411. doi:10.1177/24741264231178377

